Why Multi-Enzyme Hydrolysis Is Transforming Functional Peptide Production
In the booming global market for functional peptides—ranging from anti-aging collagen boosters to muscle-repairing sports supplements—the push for higher performance and cleaner processes is relentless. Traditional hydrolysis methods using single enzymes often fall short. They may leave behind bitter-tasting peptides, low activity fractions, or inefficient yields.
Multi-enzyme hydrolysis offers a powerful solution. By carefully designing enzyme blends that act sequentially or synergistically, manufacturers can unlock unique peptide fingerprints, achieve better functional performance, and scale production efficiently—all while meeting global clean-label and sustainability demands.
What Is Multi-Enzyme Hydrolysis?
Multi-enzyme hydrolysis refers to the use of two or more enzymes—either simultaneously or in a controlled sequence—to break down proteins into functional peptides. Unlike single-enzyme systems, which may cleave only specific peptide bonds, enzyme cocktails combine endopeptidases (which cleave internal peptide bonds) and exopeptidases (which cleave terminal amino acids) to produce smaller, highly bioactive peptides [1].
This method enables manufacturers to tailor the hydrolysis process to a target molecular weight distribution, minimize bitterness, and optimize the bioactivity of the final peptide product.
Key Parameters in MEH Process Design
Designing an effective MEH system requires precise control over several parameters:
- pH and temperature: Each enzyme in the cocktail has an optimal operating range. For example, Alcalase® functions best at pH 8.0–10.0, while Flavourzyme® prefers mildly acidic to neutral conditions.
- Enzyme-to-substrate ratio (E/S): Adjusting E/S affects the degree of hydrolysis (DH), reaction kinetics, and final yield.
- Hydrolysis time and sequence: In sequential MEH, enzymes are added at different stages, allowing better control over peptide structure.
- Mixing and aeration: These influence enzyme contact efficiency and hydrolysis consistency at scale.
Real-time monitoring using DH%, viscosity, and peptide size distribution is critical for ensuring process stability and product consistency [2].
Designing Enzyme Cocktails for Functional Peptide Production
Selecting the Right Proteases
Common enzymes used in MEH include:
- Alcalase® (from Bacillus licheniformis) – robust endoprotease, ideal for initial breakdown.
- Flavourzyme® (from Aspergillus oryzae) – mix of exopeptidases that reduces bitterness and generates bioactive peptides.
- Papain – plant-derived enzyme effective at broad pH ranges.
- Bromelain and Ficin – plant-based enzymes used for gentle hydrolysis, especially in food-grade applications.
The combination depends on the substrate—whether fish collagen, soy protein, or whey protein concentrate.
Compatibility by Substrate
| Substrate | Enzyme Combination | Target Benefit |
|---|---|---|
| Fish Collagen | Alcalase + Flavourzyme | Tripeptide yield, bioactivity |
| Soy Protein Isolate | Papain → Flavourzyme (sequential) | Bitterness reduction, ACE-I |
| Whey Permeate | Trypsin + Ficin | High solubility, rapid uptake |
These enzyme systems are optimized to release bioactive sequences such as Gly-Pro-Hyp (collagen-stimulating), Val-Pro-Pro (antihypertensive), and Leu-Lys-Pro (immune-modulating) [3].
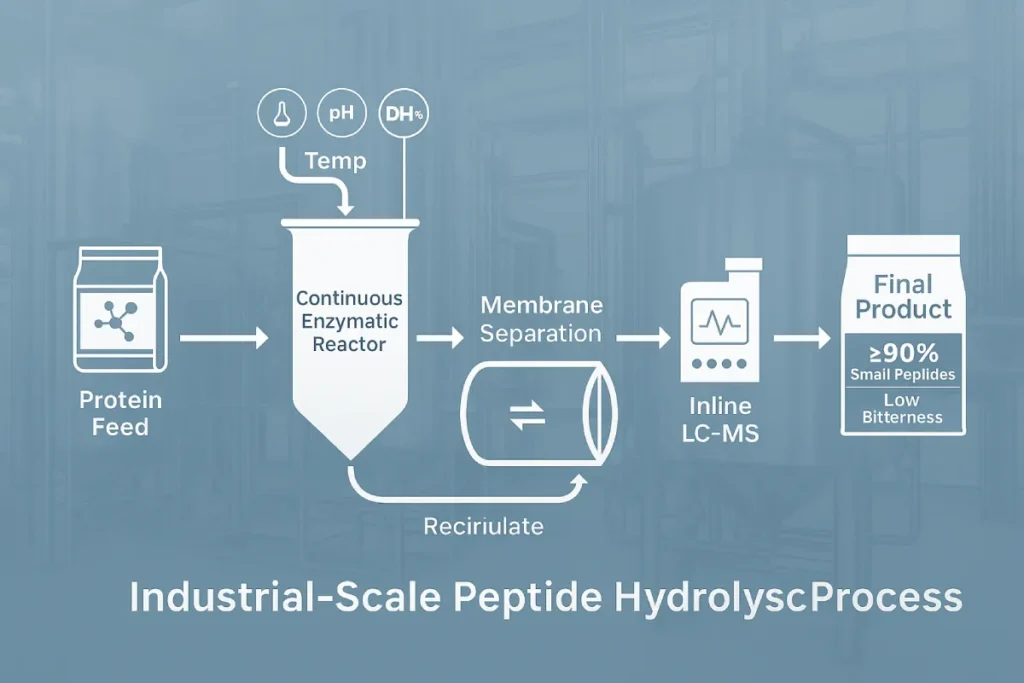
From Lab Bench to Industrial-Scale Hydrolysis
Scaling MEH from lab to commercial volume involves significant bioprocess engineering:
- Reactor type: Fed-batch and continuous plug-flow reactors are preferred over batch systems for consistent enzyme exposure and higher throughput.
- Membrane separation integration: Inline ultrafiltration enables the separation of peptides below 1 kDa while recycling larger fragments for further hydrolysis.
- Inline quality control: Peptide profiles can be monitored via LC-MS, FTIR, and near-infrared spectroscopy for real-time feedback.
For instance, at PEPDOO®, a proprietary MEH process using fish-derived collagen and dual enzymes produces nano-collagen peptides with ≥90% tripeptide content and 99.7% purity in a 3,000 L continuous stirred tank reactor (CSTR).
Upstream Preparation Matters
Functional peptide yield is influenced by how well the raw material is pretreated. For example:
- Defatting improves enzyme access in oilseed meals (e.g., soy, sea buckthorn).
- Denaturation via mild heat exposes peptide bonds for enzymatic access.
- Particle-size reduction enhances surface area for reaction kinetics.
Additionally, sourcing sustainable raw materials—from fish skin by-products to dairy permeates—adds value and aligns with circular bioeconomy practices [4].
Downstream Purification & Finishing
After enzymatic hydrolysis, downstream processes include:
- Enzyme inactivation (via heat or pH shift)
- Membrane-based fractionation for molecular weight cut-off control
- Deodorization and decolorization (e.g., with activated carbon)
- Drying using spray drying or freeze-drying to preserve peptide integrity
These steps determine product quality, solubility, color, taste, and shelf stability—critical for acceptance in premium applications like cosmeceuticals or infant nutrition.
Functional & Sensory Benefits of MEH Peptides
- Higher bioavailability: Peptides <1,000 Da are more easily absorbed in the human gut [5].
- Enhanced functionality: Specific sequences promote skin repair, muscle synthesis, or immune regulation.
- Reduced bitterness: Flavourzyme and exopeptidase activity significantly reduce off-notes often found in single-enzyme hydrolysates.
Case Study Highlights
| Product Type | Enzyme Strategy | Output |
|---|---|---|
| Marine Collagen Peptide | Alcalase + Flavourzyme | 99.7% purity, ≥90% tripeptide |
| Soy Peptide Powder | Papain → Flavourzyme (seq.) | 75% peptides <1 kDa, low bitterness |
| Dairy Peptide Isolate | Trypsin + Membrane Reactor | 4× throughput, energy saving 22% |
Quality, Regulatory & Global Trade Considerations
B2B buyers and regulatory stakeholders demand:
- Compliance with ISO 22000, FSSC 22000, HACCP, and country-specific standards (e.g., China Blue Hat, EU Novel Food).
- Certifications like Halal, Kosher, GMO-free.
- Label accuracy: hydrolyzed protein vs peptide concentrate vs bioactive tripeptide claim.
Sustainability Advantage
Compared to acid hydrolysis, MEH offers:
- 50–80% lower energy usage
- No need for neutralization or post-pH correction
- Reduced water demand
- Better protein-to-peptide conversion efficiency
Further innovations like immobilized enzymes and continuous membrane reactors improve lifecycle performance and cost efficiency [6].
Future Outlook: AI, Biotech & Custom Peptides
- AI-driven peptide prediction, enabling reverse-design of enzyme cocktails.
- CRISPR-engineered enzymes with tailored cleavage specificity.
- Fermentation-integrated systems that produce bioactive peptides alongside postbiotics or probiotics.
Conclusion
Multi-enzyme hydrolysis is a robust, scalable, and customizable strategy that enables the production of high-quality functional peptides. For manufacturers and B2B brands seeking bioactive, clean-label ingredients with strong consumer appeal, MEH offers both a technical and commercial edge.
Looking for a Custom Peptide Solution or Pilot-Scale MEH Support?
Our peptide innovation team offers feasibility studies, technical dossiers, and factory tours. Let’s discuss how we can support your next project!
Request Technical Support or Factory VisitWe’ll get back to you within 24 hours—guaranteed!
FAQ
MEH allows for synergistic cleavage of complex protein structures, yielding peptides with a narrower molecular weight distribution, improved bioactivity, and significantly reduced bitterness. It also enables higher protein conversion rates and greater control over peptide functionality—especially in large-scale reactors where uniform hydrolysis is critical. This approach is particularly valuable for developing clean-label, high-purity peptide ingredients tailored to cosmetic, medical, or functional food applications.
Different proteases cleave proteins at specific sites, producing unique peptide sequences. For instance, Alcalase® efficiently initiates hydrolysis, while Flavourzyme® refines the peptide profile and reduces bitterness. The right enzyme cocktail can enhance the generation of key bioactive motifs (e.g., collagen-boosting tripeptides or ACE-inhibitory dipeptides), while improving solubility and taste—key factors for both oral supplements and functional beverages.
Yes. MEH systems can be tailored to each substrate by adjusting enzyme type, sequence, pH, temperature, and hydrolysis time. For example, marine collagen benefits from Alcalase + Flavourzyme to achieve high tripeptide purity, while soy protein may require sequential Papain → Flavourzyme hydrolysis to optimize yield and reduce bitterness. A customized approach ensures maximum value extraction and targeted biofunctionality from each protein source.
Industrial MEH facilities implement rigorous inline process monitoring, including:
Degree of hydrolysis (DH%) tracking
Real-time molecular weight profiling using LC-MS or NIR
Membrane-based fractionation for precision separation
Batch logs & HACCP compliance
These controls ensure reproducibility, batch-to-batch consistency, and meet international quality standards such as ISO 22000 or FSSC 22000, which are essential for export and regulatory approvals.
Absolutely. MEH offers exceptional control over peptide size and sequence, enabling the production of high-purity, low-allergen, and biologically active peptides. These properties are crucial for demanding applications such as:
Nutricosmetics (e.g., tripeptide-rich collagen for skin elasticity)
Sports recovery (e.g., branched-chain or antioxidant peptides)
Medical food (e.g., hypoallergenic hydrolysates for digestion-impaired patients)
Moreover, MEH peptides can be formulated into RTDs, powders, capsules, or gummies, offering excellent formulation flexibility.
Key considerations include:
Food Safety Certifications: ISO 22000, FSSC 22000, HACCP, GMP
Cultural Certifications: Halal, Kosher, Non-GMO
Regulatory Pathways:
EU: Novel Food (EU Regulation 2015/2283)
China: Blue Hat filing for health foods
USA: GRAS notification for dietary supplements
Trusted manufacturers like PEPDOO® provide full documentation, technical dossiers, and can support regulatory filings in multiple regions.
While enzyme costs are initially higher, MEH’s higher conversion yield, reduced energy usage, and lower waste production (compared to acid or alkali hydrolysis) result in lower total cost per kg peptide. Modern MEH systems also enable continuous production with inline filtration, which increases throughput by 2–4× while lowering labor and utility costs.
Yes, many advanced peptide manufacturers offer OEM & ODM collaboration models, including:
Raw material screening
Enzyme protocol optimization
Pilot-scale trials
Stability & bioactivity testing
Regulatory dossier support
Through co-development, you can launch differentiated, IP-backed functional peptides with faster go-to-market and stronger branding.
References
- Adler-Nissen, J. (1986). Enzymic Hydrolysis of Food Proteins. Springer.
- Kristinsson, H. G., & Rasco, B. A. (2000). Fish protein hydrolysates: Production, biochemical, and functional properties. Critical Reviews in Food Science and Nutrition, 40(1), 43–81.
- Hartmann, R., & Meisel, H. (2007). Food-derived peptides with biological activity: from research to food applications. Current Opinion in Biotechnology, 18(2), 163–169.
- Bleakley, S., & Hayes, M. (2017). Algal proteins: Extraction, application, and challenges concerning production. Foods, 6(5), 33.
- Udenigwe, C. C., & Aluko, R. E. (2012). Food protein-derived bioactive peptides: production, processing, and potential health benefits. Journal of Food Science, 77(1), R11–R24.
- Rao, P. V., & Ghosh, M. (2021). Enzyme immobilization strategies and their applications in bioprocess development. Biotechnology Reports, 32, e00668.