Why Peptide Stability Is a Critical Factor in Product Success
As peptides gain traction across functional nutrition, cosmeceuticals, and pharmaceutical-grade supplements, their stability has become a decisive factor in determining product viability. For R&D teams, ensuring peptide formulation stability is not just a scientific hurdle—it’s a commercial imperative. Poorly stabilized peptides can degrade during storage, lose bioactivity, or exhibit inconsistent results, undermining both regulatory compliance and consumer trust.
In this article, we delve into the multifaceted world of peptide stability—from degradation mechanisms to formulation techniques—providing science-backed insights for formulators, procurement managers, and product developers working with functional peptide ingredients.
Why Peptide Stability Is Crucial
Peptides are short chains of amino acids known for their targeted bioactivity, high absorption rates, and minimal immunogenicity. However, their delicate molecular structure makes them prone to degradation under environmental and processing stress. In B2B peptide product development, stability is tightly linked to shelf life, batch-to-batch consistency, packaging decisions, and global regulatory approval.
Whether you are designing a collagen peptide sachet, a cognitive peptide capsule, or a functional peptide drink, the ability to maintain potency over time directly affects efficacy and ROI. For manufacturers and brands alike, understanding and improving peptide formulation stability is key to scaling a winning product.
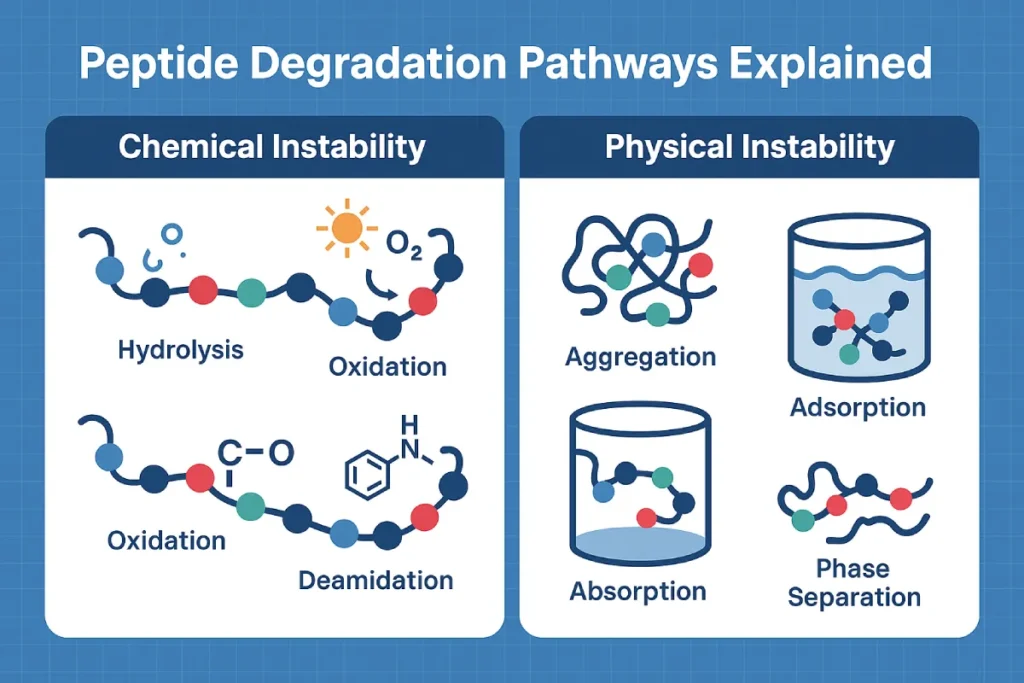
Core Degradation Mechanisms in Peptide Products
Peptides are susceptible to both chemical and physical degradation, often concurrently. The most common pathways include:
Chemical Instability
- Hydrolysis – Cleavage of peptide bonds due to moisture, pH shifts, or enzymatic activity.
- Oxidation – Sensitive amino acids such as methionine, cysteine, and tryptophan can oxidize in the presence of air, light, or metal ions [1].
- Deamidation – Asparagine and glutamine residues may undergo hydrolysis, changing their charge and disrupting structure.
- Racemization – L-to-D isomerization alters peptide bioactivity and can generate immunogenic fragments.
Physical Instability
- Aggregation – Especially common in high-concentration peptide formulations, leading to reduced bioavailability.
- Precipitation and phase separation – Often caused by pH shifts or improper solvent selection.
- Adsorption – Peptides may bind to packaging materials or container walls, resulting in dosing inconsistencies [2].
Factors That Influence Peptide Stability
Intrinsic Factors
- Amino acid sequence and length: Shorter peptides may degrade faster; charged or hydrophobic residues are more vulnerable to oxidation.
- Molecular weight and 3D structure: Cyclic or helical peptides often show improved resistance to degradation [3].
Formulation-Dependent Factors
- pH and buffer systems: Each peptide has an optimal pH range. Buffers like citrate or phosphate can stabilize structure, but require compatibility checks.
- Excipients: Sugars like trehalose and mannitol can act as stabilizers, while antioxidants (e.g., ascorbic acid) counteract oxidation.
- Solubility and ionic strength: Poorly soluble peptides are more prone to precipitation.
Environmental Stressors
- Temperature: Elevated storage or processing temperatures accelerate hydrolysis and oxidation.
- Humidity and moisture: Especially critical in peptide powders and sachets.
- Light: UV exposure can break aromatic amino acid bonds, requiring UV-protective packaging [4].
- Mechanical stress: Shaking, mixing, or freeze-thaw cycles can trigger aggregation in sensitive formulations.
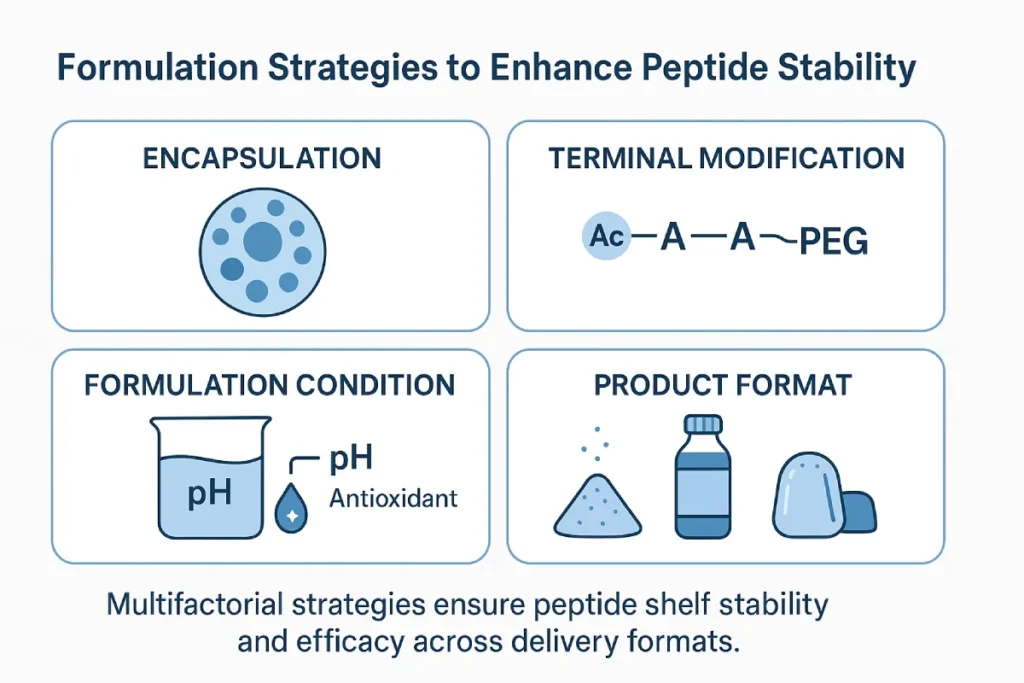
Strategies to Improve Peptide Stability
Peptide Structure Optimization
- Terminal modifications: Acetylation or amidation protects the N- and C- termini from enzymatic degradation.
- Cyclization: Enhances resistance to proteolysis.
- PEGylation or lipidation: Adds steric protection and improves solubility in complex matrices [5].
Innovative Delivery Systems
- Microencapsulation: Using liposomes, PLGA microspheres, or spray-dried capsules to protect peptides from oxidation and enzymatic degradation.
- Nanocarriers: Solid lipid nanoparticles (SLNs) and nanogels can enable targeted delivery and enhance protection in aqueous or gastrointestinal environments.
- Excipient pairing: Trehalose, lecithin, and polymeric film coatings improve dispersibility and prevent aggregation.
Formulation Formats
- Lyophilized powders: Ideal for high-purity, injectable, or reconstitutable peptide formulations.
- Sachets and tablets: Low water activity and optimized pH conditions increase longevity.
- Functional foods: Peptides integrated into bars or gummies must consider Maillard reactions and pH-activity compatibility.
Analytical Tools for Stability Evaluation
- HPLC & LC-MS/MS: Gold standards for assessing peptide purity, degradation products, and profile shifts.
- FTIR and Circular Dichroism (CD): For monitoring structural changes in secondary or tertiary conformations.
- Differential Scanning Calorimetry (DSC): Reveals thermal stability transitions in peptide matrices.
- Stability Studies per ICH Guidelines:
- Long-term (25°C/60% RH)
- Accelerated (40°C/75% RH)
- Stress testing (light, freeze-thaw, oxidation)
Predictive modeling tools are increasingly used to estimate peptide shelf life across formulation types and climates [6].
Regulatory Considerations for Stable Peptide Products
- Purity and identity: Free from unintended degradation products.
- Label claims: Accurate dosage and shelf-life must be validated.
- Stability data: Required for FDA (USA), EFSA (Europe), and SAMR (China) submissions.
GMP-compliant manufacturing and validated QA protocols are essential for international peptide trade and brand reputation.
Real-World Case Studies
- Collagen Peptide Drink (pH-adjusted): Reformulating to pH 4.2 with ascorbic acid extended shelf life by 12 months without preservatives.
- Cognitive Peptide Capsule: Cyclization of key nootropic peptide increased storage stability under high humidity by 42%.
- Peptide Bar Application: A functional sports peptide blend was successfully embedded in a protein bar matrix using trehalose + lecithin, maintaining >90% bioactivity after 6 months.
Best Practices for R&D and Scale-Up
- Early integration of stability studies in product pipeline planning.
- Cross-functional team alignment: R&D, QC, packaging, and marketing must collaborate.
- Pilot batch testing under real-world conditions.
- Data-driven decisions using accelerated testing and modeling.
Peptide stability is not a final checkpoint—it must be engineered into the core of formulation development.
Future Outlook: AI and Peptide Formulation
- AI-driven peptide stability predictions based on sequence and environmental inputs.
- Machine-learning models to screen excipient-peptide compatibility faster.
- Digital twins for in silico simulation of shelf life across packaging and logistics scenarios.
Conclusion: Stability Is the Foundation of Success
Peptide formulation stability is where product efficacy, customer satisfaction, and market compliance intersect. From collagen supplements to metabolic peptide blends, the ability to maintain integrity and performance over time is essential. Whether you’re a brand formulator, OEM partner, or procurement head, now is the time to prioritize peptide stability in every product decision.
🧪 Seeking a Full-Process Peptide Manufacturing Partner?
With in-house enzymatic hydrolysis, encapsulation, and quality assurance,
PEPDOO® delivers formulation-ready peptides backed by stability data and global technical support.
Request a sample or collaborate on your next custom R&D project today.
Our formulation experts will reply within 1 business day.
FAQ
Peptides are prone to chemical degradation such as hydrolysis, deamidation, oxidation, and isomerization, especially in water-based systems. Physical instability, including aggregation and adsorption, also accelerates deterioration.
Recommended solutions: pH optimization, addition of antioxidants/chelators, oxygen exclusion, and temperature control (e.g., cold-chain logistics).
Asp-X or Asn-X sequences: prone to deamidation and cyclic imide formation.
Cysteine and methionine: highly oxidation-sensitive.
N-terminal glutamine: may cyclize into pyroglutamate, altering identity and purity .
Avoid sequences with Asp-Pro, Asn-Gly, or N-terminal Gln.
Replace sensitive residues (e.g., Met → Norleucine, Cys → Ser).
Keep hydrophobic residues below 50%; include at least one charged residue every five amino acids to enhance solubility.
Minimize stretches of Pro or Ser to reduce risk of isomerization .
Forced degradation studies simulating pH, heat, light, oxidation stress.
Comprehensive analytics: UPLC-MS, HPLC, ThioT, DLS to assess both chemical and physical stability.
Predictive modeling: Machine learning (e.g., RF, MLP) can anticipate long-term degradation, enabling faster formulation cycles .
pH and buffer optimization to reduce degradation pathways.
Addition of stabilizers like polyols (mannitol, sucrose), amino acids, PEG.
Oxygen removal or nitrogen purging to prevent oxidation.
Viscosity enhancement using polymers or carriers to minimize aggregation .
🌀 Cyclization (e.g., head-to-tail or side-chain loop): improves protease resistance.
🔁 D-amino acid or non-natural substitutions: enhance enzymatic stability.
🛡️ PEGylation, lipidation, glycosylation: reduce renal clearance and prolong plasma half-life .
Lyophilized (dry powder) format is preferred over liquid to minimize hydrolysis.
Cold storage (2–8°C or −20°C) is typically recommended.
Aliquoting and avoiding repeated freeze-thaw cycles help preserve integrity.
References
- Wang, W. (2005). Instability, stabilization, and formulation of liquid protein pharmaceuticals. International Journal of Pharmaceutics, 289(1-2), 1–30.
- Zhang, Y., & Creagh, A. L. (2020). Strategies to prevent peptide adsorption in pharmaceutical formulations. Pharmaceutical Research, 37(4), 85.
- Chatterjee, J., Rechenmacher, F., & Kessler, H. (2013). N-methylation of peptides and proteins: an important element for modulating biological functions. Angewandte Chemie International Edition, 52(1), 254-269.
- Mahler, H. C., Friess, W., Grauschopf, U., & Kiese, S. (2009). Protein aggregation: pathways, induction factors and analysis. Journal of Pharmaceutical Sciences, 98(9), 2909–2934.
- Fosgerau, K., & Hoffmann, T. (2015). Peptide therapeutics: current status and future directions. Drug Discovery Today, 20(1), 122–128.
- Yu, L. X. (2008). Pharmaceutical quality by design: product and process development, understanding, and control. Pharmaceutical Research, 25(4), 781–791.